Concept
Our research is based on utilizing the synergy of functional groups to develop new high-performance polymers and to explore new reactivity for polymer synthesis and degradation. The details of this concept are summarized in our recent review.
Topic-1| Conjugate Substitution
- Robustness: Operable under ambient conditions, even in aqueous media or in the solid state.
- Rapidity: Completed within a few minutes in the presence of a suitable catalyst.
- Convenience: When the leaving group is a halogen atom, the byproduct can be removed by simple washing with water.
- Versatility: Various nucleophiles, such as carboxylic acids, phenols, thiols, and amines, can be used.
- Controllability: The reaction can be reversible or irreversible depending on the choice of nucleophiles and leaving groups.
Conjugate substitution is a reaction between allyl-substituted methacrylic compounds and nucleophiles, characterized by:
We explore the polymer chemistry using conjugate substitution reaction
(1) Monomer Synthesis
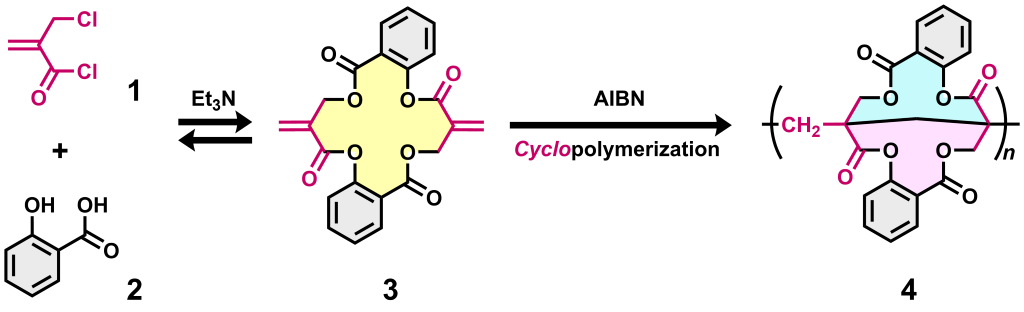
Conjugate substituion is a powerful tool in monomer synthesis. In our recent report, we investigated the condensation of 2-(chloromethyl)acryloyl chloride (1) and salicylic acid (2), based on acyl and conjugate substitutions. The 2-(acyloxymethyl) methacrylate moiety in the product undergoes reversible bond exchange via conjugate substitution in the presence of a tertiary amine catalyst, resulting in the formation of the thermodynamically stable cyclic dimer 3 in high yield. Radical polymerization of 3 resulted in cyclopolymerization (annulation polymerization) to afford acrylic polymer 4 composed of bridged bicyclic units, exhibiting high Tg and good solubility.
(2) Polymerization and Degradation
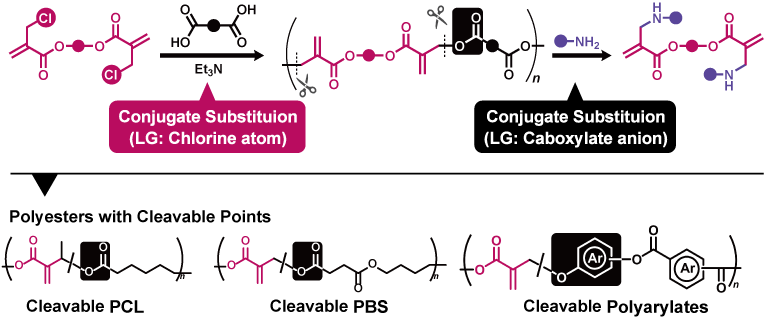
Unsaturated polyesters containing acrylic moieties are thermally unstable, which limits their synthetic accessibility. In contrast, conjugate substituion proceeds at ambient temperature, eliminating concerns about thermally induced side reactions. Therefore, polycondensation using conjugate substituion enables the facile synthesis of unsaturated polyesters.
Conjugate substitution is also effective for polymer degradation. For example, the unsaturated polyesters obtained as described above undergo backbone cleavage via conjugate substitution with amines. This reaction proceeds not only in solution but also in the solid state and in aqueous suspensions, making it promising for applications in materials science. In fact, we have also developed a method to introduce acrylic moieties as cleavable points into conventional polyesters.
(3) Vitrimers
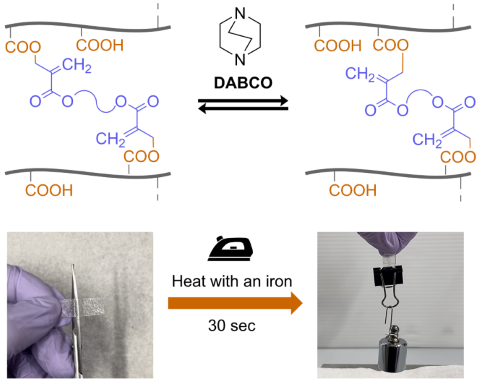
In crosslinked polymers, the mobility of polymer chains is highly restricted, making melt processing and stress relaxation difficult. In contrast, recent studies have shown that introducing exchangeable bonds (dynamic covalent bonds) at the crosslinking points enables network rearrangement, thereby imparting stress relaxation, processability, and self-healing properties.
In our laboratory, we have developed crosslinked acrylic elastomers (vitrimer-like materials) based on bond exchange via conjugate substitution. These elastomers exhibit orders-of-magnitude faster stress relaxation compared to analogous acrylic-based vitrimers, allowing for reprocessing even with a household iron and offering potential applications as adhesive sheets. Furthermore, a variety of materials can be produced by selecting different base polymers, such as crosslinked acrylic glass based on methacrylic polymers.
Topic-2 | Cleavable Vinyl Polymers
Vinyl polymers are highly attractive materials due to the wide variety of available monomers, the ability to tune their properties through copolymerization, and the ease of synthesis via photopolymerization. However, because their main chains consist of chemically stable carbon–carbon backbones, they are generally difficult to degrade, posing a challenge for sustainability. In our laboratory, we address this issue by exploiting the synergistic effects of functional groups to develop degradable vinyl polymers.
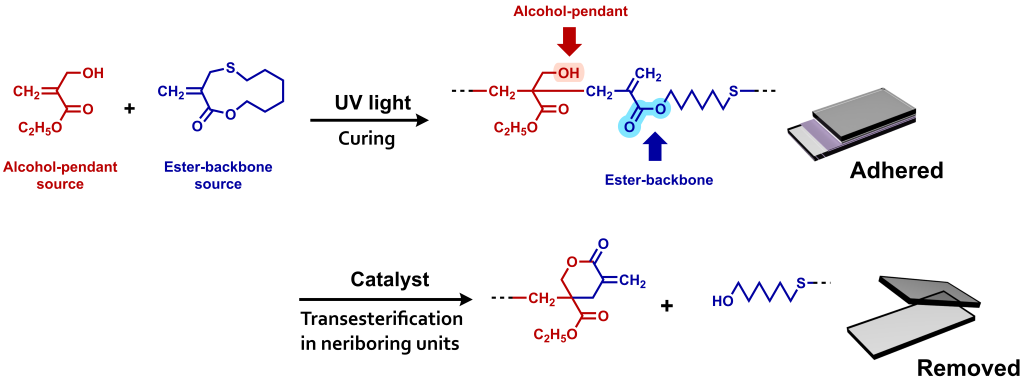
(1) Removable Adhesives
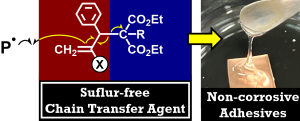
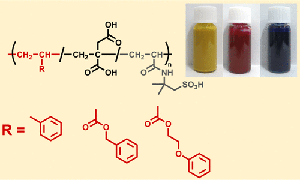
Introducing ester linkages into the main chain via radical ring-opening polymerization imparts degradability to acrylic polymers. However, since ester groups are also present in the side chains, selectively cleaving only the main chain is not straightforward. In our laboratory, we have developed a strategy to achieve selective main-chain degradation by employing acrylic monomers bearing alcohol functionalities, which effectively promote transesterification between the side chains and the main chain. Furthermore, this concept is advanced to UV-curable adhesives with controlled removability.
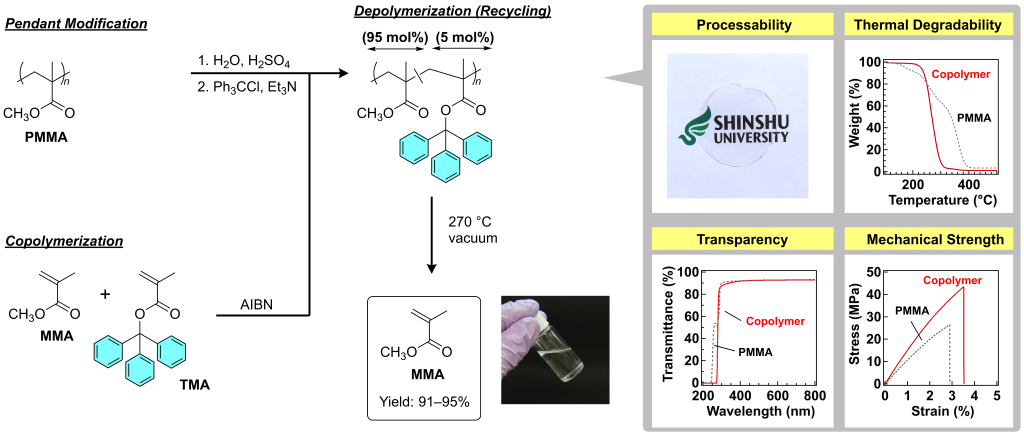
(2) Chemical Recycling
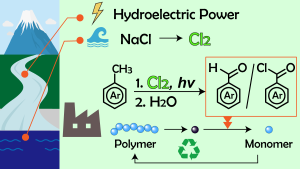
Poly(methyl methacrylate) (PMMA) is commnly used in our dialy life as acrylic glasses. Recently, we reported modified PMMA with excellent recyclability. This methacrylic polymers, bearing trityl ester pendants, exhibit optical and mechanical properties comparable to prestine PMMA, whereas it recovers the monomer (MMA) via heating at 270 ° C
(3) Recyclable Vinyl Polymers
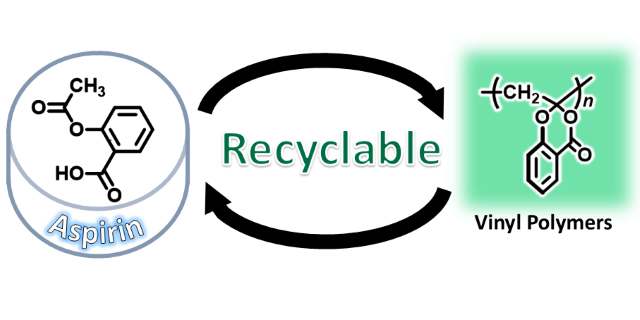
We propose new molecular design for recycable vinyl polymers. For example,the vinyl polymers derived from acetylsalicylic acid, so-called aspirin, is decomposed to salicylic acid and acetic acid via hydrolysisWe also reportedpolystyrene analogues that undergo depolymerization triggered by pendant hydrolysis.